
Dipole moment formula charge on the atoms the distance between them D Q × R. Hence, it causes a non-zero dipole moment which causes the polar nature of PF3. 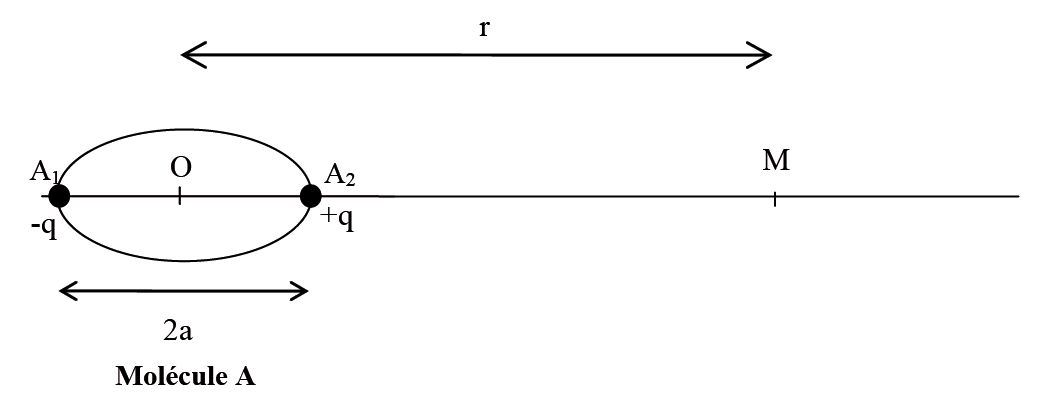
In the case of NH 3, the orbital dipole due to the lone pair is in the same direction as the resultant dipole moment of the N H bonds. The dipole of the PF3 molecule originates in the downward and upward direction because of the lone pair present in Phosphorous. For some highly symmetrical structures, the individual bond dipole moments cancel one another, giving a dipole moment of zero. Which of the following pairs of molecules is listed in order of decreasing net dipole moment (from more polar molecule to less polar molecule) A. As the electronegativity of nitrogen (3.04) is greater than that of hydrogen (2.2) the result is that the N-H bonds are polar with a net negative charge on the nitrogen atom and a smaller net positive charge on the hydrogen atoms. I cannot connect the separation of positive and negative charges definition with lone pair. But I am not getting the intuition why and how lone pairs affect the polarity and dipole moment. the seperation of positive and negative charges. In other words, they are polar molecules. Lone pairs can make a contribution to a molecules dipole moment. Dipole moment is the degree of polarity, i.e. 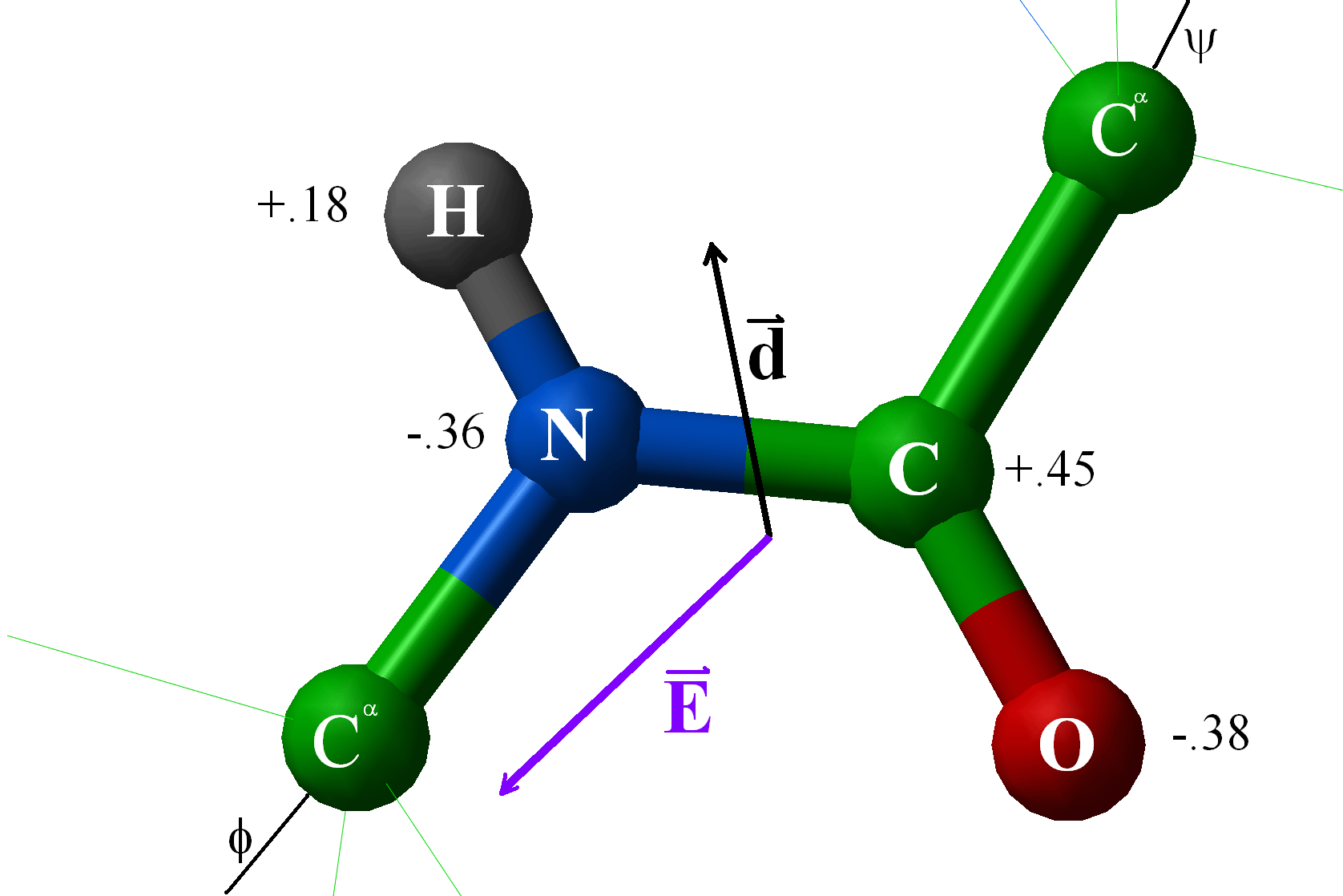 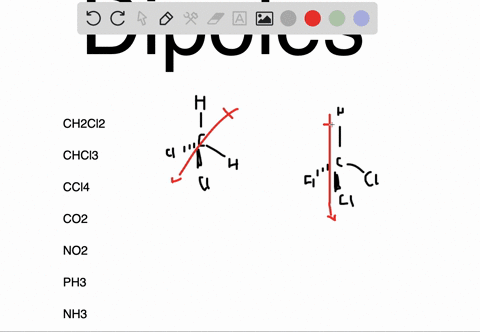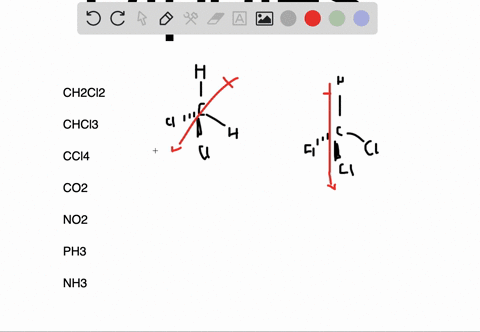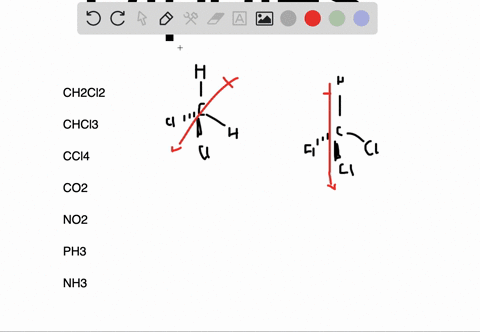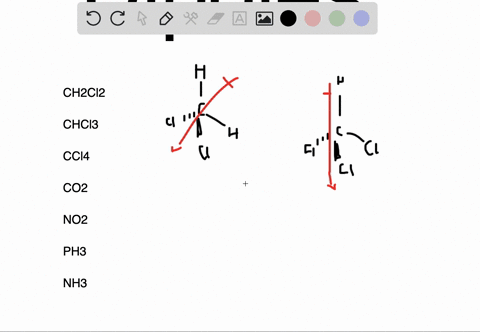
Molecules such as HF, HCI, NH H O NF etc., have polar bonds and their 3' dipole moment is greater than zero. The dipole moment of lone pairs in $N$C m.A molecule may be nonpolar either when there is an equal sharing of electrons between the two atoms of a diatomic molecule or because of the symmetrical arrangement of polar bonds in a more complex molecule. It can be defined as the product of the distance between the centers of a positive and negative charge, and the magnitude of the charge. Any diatomic molecule with a polar covalent bond has a dipole moment, but in polyatomic molecules, the presence or absence of a net dipole moment depends on the structure. Molecules such as C02, BF3, CH4, CC14, PC15, SF6, etc., have polar bonds, but they are nonrpoar molecules because of their zero value of dipole moment.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
